Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Yamashita, Shinichi; Hiroki, Akihiro; Nagasawa, Naotsugu; Murakami, Takeshi*; Taguchi, Mitsumasa
no journal, ,
A new polymer gel dosimeter was developed to overcome weakness of earlier polymer gel dosimeters. Hydroxyethyl 2-methacrylate (HEMA) and polyethylene glycol ester dimethacrylate (9G) were less toxic constituents, which polymerize with irradiations. One of cellulose derivatives, hydroxypropyl cellulose (HPC), was crosslinked by irradiations of electron beam and used as a gel matrix, which doesn't soften with increasing temperature. Hydrogel of HPC is very transparent, so light scattering and absorption of products of radiation-induced polymerization are easily observable. As a result of irradiation, white turbidity was observed inside the gel at around 1-2 Gy. Increasing behavior of white turbidity with irradiation depended on composition of constituent monomers. White turbidity appears at rather lower dose and its increase saturates at around 10 Gy of 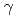 -irradiation when 9G is predominant. On the other hand, it becomes stronger even beyond 10 Gy when HEMA is predominant. In order to quantify the degree of white turbidity, haze measurement and UV-vis absorption spectrometry were conducted. In addition, white turbidity evolution was also confirmed after irradiations of therapeutic C ion beam although degree of white turbidity was less clear than that observed after
-irradiation when 9G is predominant. On the other hand, it becomes stronger even beyond 10 Gy when HEMA is predominant. In order to quantify the degree of white turbidity, haze measurement and UV-vis absorption spectrometry were conducted. In addition, white turbidity evolution was also confirmed after irradiations of therapeutic C ion beam although degree of white turbidity was less clear than that observed after  Co
Co 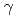 -irradiation.
-irradiation.
 produced in water radiolysis with
produced in water radiolysis with  Co
Co 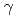 -rays and C ion beams
-rays and C ion beamsYamashita, Shinichi; Katsumura, Yosuke; Murakami, Takeshi*
no journal, ,
Main initial products of water radiolysis are e
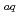 and
and  OH, which go through intraspur/track reactions in the time regime from ps to
OH, which go through intraspur/track reactions in the time regime from ps to 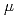 s, leading to production of H
s, leading to production of H O
O and H
and H . Among the main products, only H
. Among the main products, only H is not soluble in water and difficult to be accurately quantified. H
is not soluble in water and difficult to be accurately quantified. H also tends to leak even from a very small crevice where other gases do not leak. It is necessary to pay attentions to leakage and equilibrium between liquid-gas phases when ones want to accurately quantify H
also tends to leak even from a very small crevice where other gases do not leak. It is necessary to pay attentions to leakage and equilibrium between liquid-gas phases when ones want to accurately quantify H . Therefore, an online analysis apparatus of gaseous products arising from liquid samples has been developed to measure H
. Therefore, an online analysis apparatus of gaseous products arising from liquid samples has been developed to measure H production from water. In this work, at first accuracy and sensitivity of the apparatus were improved, and then, H
production from water. In this work, at first accuracy and sensitivity of the apparatus were improved, and then, H yields were measured as a function of concentration of the nitrate anion, which is a scavenger not only for e
yields were measured as a function of concentration of the nitrate anion, which is a scavenger not only for e
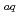 but also for the pre-solvated electron (e
but also for the pre-solvated electron (e
 ). Figure shows G(H
). Figure shows G(H ) measured in this work in comparison with reported values. Well-established primary G value for
) measured in this work in comparison with reported values. Well-established primary G value for  Co
Co 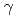 -irradiation, 0.46 (100 eV)
-irradiation, 0.46 (100 eV) , is obtained at low NO
, is obtained at low NO
 concentrations. Our data with
concentrations. Our data with  Co
Co 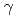 -irradiation were in good agreement with the reported values. In the cases of C ion irradiations G(H
-irradiation were in good agreement with the reported values. In the cases of C ion irradiations G(H ) values increased with increasing LET due to increasing significance of intratrack reactions. Further discussion on H
) values increased with increasing LET due to increasing significance of intratrack reactions. Further discussion on H production in fast process before 1 ps necessitate measurements of G(H
production in fast process before 1 ps necessitate measurements of G(H ) at other NO
) at other NO
 concentrations and with the usage of other scavengers of e
concentrations and with the usage of other scavengers of e
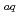 and e
and e
 .
.
Kimura, Atsushi; Nagasawa, Naotsugu; Taguchi, Mitsumasa
no journal, ,
no abstracts in English
Hiroki, Akihiro; Sato, Toshihiro*; Nagasawa, Naotsugu; Taguchi, Mitsumasa; Tamada, Masao
no journal, ,
no abstracts in English
 ray irradiation on deoxidation in aqueous solution by hydrazine
ray irradiation on deoxidation in aqueous solution by hydrazineMotooka, Takafumi; Sato, Tomonori; Yamamoto, Masahiro
no journal, ,
The effect of  ray irradiation on deoxidation in aqueous solution by hydrazine was investigated using pure water and artificial sea water at room temperature. We placed the solutions with small amount of hydrazine at
ray irradiation on deoxidation in aqueous solution by hydrazine was investigated using pure water and artificial sea water at room temperature. We placed the solutions with small amount of hydrazine at  ray irradiation field at dose rates of 0.3-7.5 kGy/h. The concentration of dissolved oxygen in the solutions was measured before and after
ray irradiation field at dose rates of 0.3-7.5 kGy/h. The concentration of dissolved oxygen in the solutions was measured before and after  ray irradiation. Concentration of dissolved oxygen was not decreased without
ray irradiation. Concentration of dissolved oxygen was not decreased without  ray irradiation, while concentration of dissolved oxygen was markedly decreased for one hour
ray irradiation, while concentration of dissolved oxygen was markedly decreased for one hour  ray irradiation, which is under detection limit at higher dose rate over 2 kGy/h. It is believed that the hydrazine addition to spent nuclear fuel pool water is effective as a method for decreasing concentration of dissolved oxygen.
ray irradiation, which is under detection limit at higher dose rate over 2 kGy/h. It is believed that the hydrazine addition to spent nuclear fuel pool water is effective as a method for decreasing concentration of dissolved oxygen.
Hanawa, Satoshi; Hata, Kuniki; Chimi, Yasuhiro; Nishiyama, Yutaka; Nakamura, Takehiko
no journal, ,
no abstracts in English
Kumagai, Yuta; Nagaishi, Ryuji; Kimura, Atsushi; Taguchi, Mitsumasa; Nishihara, Kenji; Yamagishi, Isao
no journal, ,
For decontamination of radioactive water containing salts from seawater in Fukushima Dai-ichi Nuclear Power Station, zeolite is used as adsorbent for radioactive Cs. The management of hydrogen produced by radiolysis of water in the zeolite waste is important for safe storage of the waste. Thus hydrogen production from the mixture of mordenite-type zeolite and seawater was studied. The hydrogen yield from the mixture decreased monotonically as the weight fraction of seawater was decreased. However, the measured yield was higher than the yield expected from the weight fraction of seawater, supposing that mordenite has no effect. The result suggests that hydrogen was produced in another pathway besides the direct radiolysis of seawater in the presence of mordenite but its efficiency of the hydrogen production was lower than the direct radiolysis of seawater. The radiation energy deposited to mordenite is expected to be involved in the radiolysis of the adsorbed water.
Nagaishi, Ryuji; Kondo, Takafumi*; Aoyagi, Noboru; Taguchi, Mitsumasa; Yang, J.*; Yoshida, Yoichi*
no journal, ,
Reaction behaviors of radiolysis products in mixtures of different ionic liquids containing thiocyanate anion were studied by using pulse radiolysis technique. Dimer radical ion of thiocyanate in the visible range and electron in the near-infrared range were observed in the ns-pulse radiolysis to measure their yields and reactions. The formations of products increased with increasing the thiocyanate concentration, regardless of kinds of cations in the mixture. The products were also observed in the ps-pulse radiolysis to clarify their formation pathways.
Lin, M.; Katsumura, Yosuke*; Muroya, Yusa*; Meesungnoen, J.*; Mostafavi, M.*; Jay-Gerin, J.-P.*
no journal, ,
In this invited talk, the latest results of the decay kinetics at room temperature of hydrated electron and hydroxyl radical obtained by picosecond pulse radiolysis will be introduced and then compared with the previous studies and the Monte-Carlo simulations. On the other hand, some experimental results about temperature effects on the temporal behaviors of hydrated electron obtained by direct kinetic measurements and scavenging method will be presented together with the Monte-Carlo studies on the evaluation of the time of spur expansion in low-LET high temperature radiolysis of water.
Hata, Kuniki; Hanawa, Satoshi; Kasahara, Shigeki; Muroya, Yusa*; Katsumura, Yosuke*
no journal, ,
Fukushima Daiichi Nuclear Power Units 1 - 3 experienced seawater injection into the reactor cores as an emergent measure to cool the reactors for a short period after the accident. As a result, structural materials in the reactors have been exposed to unexpected corrosion environment due to anions, such as Cl , in seawater. Furthermore, strong oxidants, such as ClO
, in seawater. Furthermore, strong oxidants, such as ClO and ClO
and ClO
 , through radiolysis of seawater by
, through radiolysis of seawater by  -rays would make the corrosion environment severer. Several studies on radiolysis of seawater at ambient temperature had been performed in the field of geological disposal of high level wastes, and amount of ClO
-rays would make the corrosion environment severer. Several studies on radiolysis of seawater at ambient temperature had been performed in the field of geological disposal of high level wastes, and amount of ClO
 and gas products had been evaluated. However, further examination is required on radiation induced reactions among seawater components to predict precisely the corrosion environment of the structural materials exposed to seawater. In the present study, the reactions of Cl
and gas products had been evaluated. However, further examination is required on radiation induced reactions among seawater components to predict precisely the corrosion environment of the structural materials exposed to seawater. In the present study, the reactions of Cl derived radicals with Br
derived radicals with Br were measured by pulse radiolysis. The production of mixed radical anion, BrCl
were measured by pulse radiolysis. The production of mixed radical anion, BrCl , was observed in aqueous solutions containing 1 mol dm
, was observed in aqueous solutions containing 1 mol dm Cl
Cl with small amounts of Br
with small amounts of Br . Model calculations are required to evaluate reaction rates of the reactions occurred in the complex solutions.
. Model calculations are required to evaluate reaction rates of the reactions occurred in the complex solutions.
Iwamatsu, Kazuhiro; Taguchi, Mitsumasa; Sugo, Yumi; Kurashima, Satoshi; Yamashita, Shinichi; Katsumura, Yosuke
no journal, ,
no abstracts in English
Nagasawa, Naotsugu; Kimura, Atsushi; Hiroki, Akihiro; Taguchi, Mitsumasa
no journal, ,
no abstracts in English
Nguyen, T. K. L.; Nagasawa, Naotsugu; Kimura, Atsushi; Hiroki, Akihiro; Taguchi, Mitsumasa
no journal, ,
The combined method of  irradiation and H
irradiation and H O
O oxidizing agent has showed the advantage in using low irradiation doses of 5-30 kGy to get a large scale of oligoalginate compare with early methods. It is concluded that low molecular weight AlgNa and/or oligoalginate can effectively be obtained by
oxidizing agent has showed the advantage in using low irradiation doses of 5-30 kGy to get a large scale of oligoalginate compare with early methods. It is concluded that low molecular weight AlgNa and/or oligoalginate can effectively be obtained by  -rays irradiation at low dose in the presence of H
-rays irradiation at low dose in the presence of H O
O .
.
Hirade, Tetsuya; Oka, Toshitaka
no journal, ,
Positronium(Ps) formation in insulating materials can be explained by spur reactions at the terminal spur of the positron track, so-called positron spur. Ps formation time is probably about 1ps and the yields of Ps formation give information of the reactions of the reactive species in the terminal spur, such as the free excess electrons. Ortho-Ps annihilation lifetime is about 1-5ns by the pick-off annihilation in condensed materials. Hence the o-Ps reactions with reactive species are quite possible. The most important reactions are, probably, ortho-Ps oxidation and spin conversion by reactive species, such as OH radical. We are introducing what we can study for water radiolysis by use of positron annihilation methods.
Katsumura, Yosuke*; Matsuura, Chihiro*; Yamashita, Shinichi; Hirade, Tetsuya; Iwamatsu, Kazuhiro; Taguchi, Mitsumasa
no journal, ,
Relevant to the nuclear accident at Fukushima Dai-ichi Nuclear Power Station, a radiolysis study of boiling water irradiated with Co-60  rays has been conducted. The evolution of hydrogen gas was clearly detected under boiling condition, while an extremely small amount of gas formation was found under the stable condition without boiling even at 97
rays has been conducted. The evolution of hydrogen gas was clearly detected under boiling condition, while an extremely small amount of gas formation was found under the stable condition without boiling even at 97 C. The G(H
C. The G(H ) of 0.37 was obtained under boiling condition. It was found that solution of 3.5% NaCl, similar to seawater, enhances the evolution, and the value of 0.7 was obtained. On the contrary, the O
) of 0.37 was obtained under boiling condition. It was found that solution of 3.5% NaCl, similar to seawater, enhances the evolution, and the value of 0.7 was obtained. On the contrary, the O gas formation was much lower than that of H
gas formation was much lower than that of H , suggesting the formed H
, suggesting the formed H O
O is not decomposed and remained in liquid. In addition, it was found that the evolved hydrogen is distributed selectively at the inner surface of the container under boiling condition. Gas injection experiments were also done without irradiation to clarify the behavior of hydrogen in the container.
is not decomposed and remained in liquid. In addition, it was found that the evolved hydrogen is distributed selectively at the inner surface of the container under boiling condition. Gas injection experiments were also done without irradiation to clarify the behavior of hydrogen in the container.